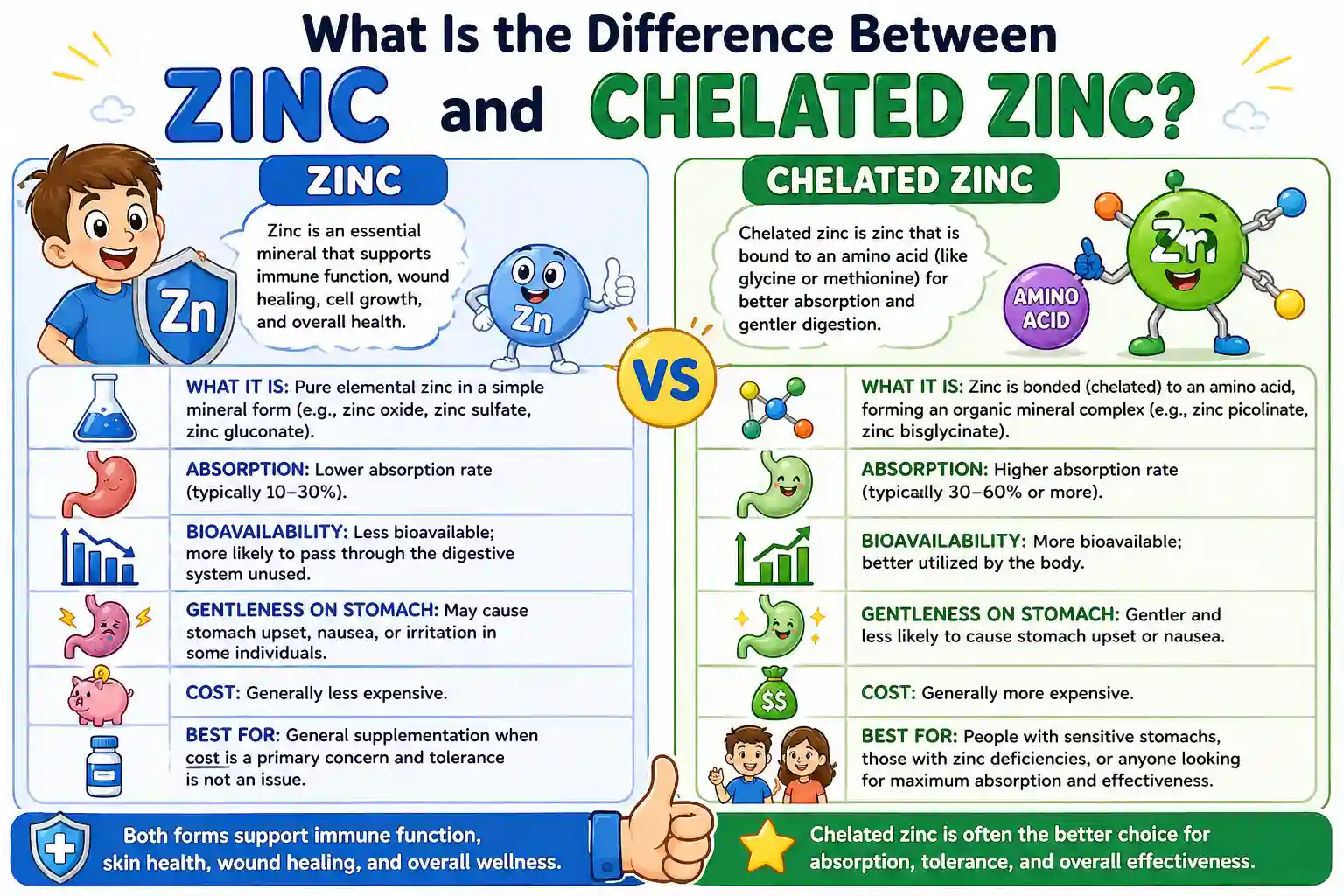
Chelated zinc is zinc bound to organic molecules like amino acids, which protects it during digestion and improves absorption, while standard zinc forms are inorganic salts that can break down early, compete with other nutrients, and are often less efficiently absorbed.
The distinction is not about the mineral itself but about how your body handles it after ingestion. Two supplements may list the same milligrams of zinc, yet deliver very different results depending on their structure.
The Absorption Barrier: Why Form Matters
Your digestive system is not designed to absorb free minerals efficiently. Zinc must pass through the intestinal lining using specialised transport proteins, mainly ZIP and ZnT transporters, which regulate how much enters and exits cells.
Standard zinc forms dissociate quickly in the stomach, releasing free zinc ions. These ions are reactive and tend to bind with other dietary compounds before they reach absorption sites.
Chelated zinc avoids this early breakdown. Because it remains attached to its ligand, it travels more intact through the digestive tract and is absorbed through alternative pathways that resemble how amino acids are taken up.
What “Chelated” Really Means at a Molecular Level
Chelation is a chemical process where a mineral is bound to an organic molecule, known as a ligand. This bond is stronger and more stable than the ionic bonds found in basic mineral salts.
The ligand acts like a carrier. Instead of leaving zinc exposed to the digestive environment, it keeps the mineral shielded until it reaches the small intestine.
This bond strength is described by stability constants. A higher stability constant indicates that the zinc remains bound longer, reducing premature reactions and improving delivery efficiency.
Why Standard Zinc Often Underperforms
Standard zinc supplements include forms like zinc oxide, zinc sulfate, and zinc gluconate. While these are widely used, their effectiveness varies significantly.
Zinc oxide, for example, contains a high percentage of elemental zinc but has relatively low solubility. This means less of it dissolves and becomes available for absorption.
Zinc sulfate dissolves more readily but is known for causing stomach irritation in some individuals. The rapid release of zinc ions can overwhelm the gastric lining, especially when taken without food.
Zinc gluconate sits somewhere in between, offering moderate absorption with better tolerance, but it still lacks the protective mechanism seen in chelated forms.
Gastric Irritation: The Real Reason Behind Zinc Nausea
Digestive discomfort is one of the most common complaints with zinc supplementation. This is often misunderstood as a sensitivity to zinc itself.
The issue lies in how the mineral behaves in the stomach. When inorganic zinc salts dissolve, they release free ions that can irritate the gastric mucosa.
This irritation can trigger nausea, especially at higher doses or when taken on an empty stomach. It is not a sign that zinc is unsuitable, but rather that the form may not be ideal.
Chelated zinc minimises this effect because it does not release zinc prematurely. The mineral remains bound, reducing direct contact with the stomach lining.
The Role of Gastric pH in Zinc Absorption
Stomach acidity plays a crucial role in mineral absorption. Standard zinc forms rely on a low pH environment to dissolve properly.
If stomach acid levels are reduced, which can happen due to age, stress, or medications like proton pump inhibitors, zinc absorption may decline.
Chelated zinc is less dependent on acidic conditions. Its stability allows it to remain intact regardless of pH variations, making it a more consistent option across different digestive states.
This is particularly relevant if you experience bloating, indigestion, or regularly use antacids.
Chelated Zinc and Amino Acid Pathways
One of the key advantages of chelation is the use of amino acid transport systems. When zinc is bound to amino acids, the body recognises it as a nutrient complex rather than a free mineral.
This allows it to be absorbed through peptide transport channels, which are generally more efficient and less competitive than mineral pathways.
It also reduces competition with other minerals such as iron and calcium, which often share the same absorption routes.
Types of Chelated Zinc and Their Differences
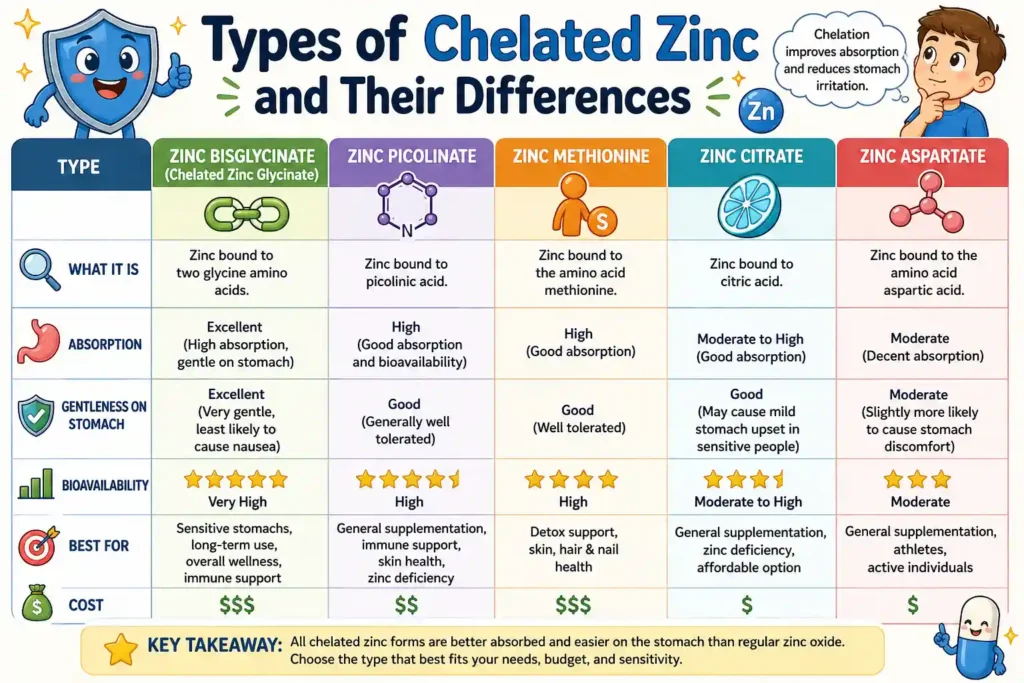
Not all chelated zinc supplements are identical. The type of ligand used influences both absorption and tolerance.
- Zinc Bisglycinate: This form is bound to two glycine molecules. It is known for high bioavailability and minimal digestive discomfort.
- Zinc Picolinate: Bound to picolinic acid, a compound naturally involved in mineral absorption. It is often used in clinical settings.
- Zinc Methionine: Attached to methionine, an essential amino acid. It is sometimes used for targeted nutritional support.
Each of these forms provides a slightly different absorption profile, but all share the advantage of improved stability.
The Antagonist Effect: Why Diet Matters
Zinc absorption is heavily influenced by what you eat. Certain compounds in food can bind to zinc and prevent it from being absorbed.
Phytates, found in whole grains and legumes, are one of the strongest inhibitors. Oxalates, present in foods like spinach, also interfere with mineral uptake.
Standard zinc forms are more vulnerable to these interactions because they exist as free ions in the digestive tract.
Chelated zinc is less affected because the ligand protects the mineral from binding with these inhibitors, allowing more of it to reach absorption sites.
Comparative Analysis: Zinc vs. Chelated Zinc
| Feature | Standard Zinc (Oxide, Sulfate, Gluconate) | Chelated Zinc (Bisglycinate, Picolinate) |
| Chemical Structure | Inorganic salt | Bound to an organic ligand |
| Absorption Pathway | Competes with minerals | Uses amino acid pathways |
| Bioavailability | Variable, often lower | Higher and more consistent |
| Gastric Tolerance | Can cause irritation | Generally gentle |
| Dependence on Stomach Acid | High | Low |
| Interaction with Food Compounds | Easily inhibited | More resistant |
Bioavailability: What the Research Suggests
Bioavailability refers to how much of a nutrient enters circulation and becomes usable by the body. It is not determined by the label dosage alone.
Some studies suggest that amino acid–chelated forms like bisglycinate can lead to higher plasma zinc levels compared to inorganic forms. This does not mean standard zinc is ineffective, but it highlights the efficiency difference.
Higher bioavailability also means you may require smaller doses to achieve the same physiological effect, which can reduce the risk of side effects.
Cost vs. Effectiveness: Looking Beyond Price
Standard zinc supplements are typically more affordable and widely available. This makes them a practical choice for short-term use or general supplementation.
Chelated zinc often costs more due to the additional processing involved in binding the mineral to organic molecules.
However, the higher absorption rate can make it more cost-effective over time. You are not just paying for zinc content, but for how efficiently your body can use it.
When Standard Zinc May Still Be Useful
Despite its limitations, standard zinc has its place. It can be effective when taken correctly and in appropriate contexts.
- Short-term supplementation during acute deficiency
- Situations where cost is a limiting factor
- Individuals who tolerate it well without digestive issues
Proper timing, such as taking it away from high-phytate meals, can improve its effectiveness.
When Chelated Zinc Becomes the Better Option
Chelated zinc is often preferred in situations where absorption or tolerance is a concern.
- Long-term supplementation for maintaining levels
- Sensitive digestive systems prone to nausea
- Diets high in fibre or plant-based foods
- Conditions affecting stomach acid production
Its stability and reduced interaction with dietary inhibitors make it a more reliable choice.
Practical Tips to Improve Zinc Absorption
Even with the right form, how you take zinc matters.
- Avoid taking zinc with coffee or tea, as these can interfere with absorption
- Separate zinc from calcium and iron supplements by a few hours
- Take zinc with a light meal if you experience stomach discomfort
- Maintain a consistent daily intake rather than irregular high doses
These small adjustments can significantly influence how well your body utilises the mineral.
Final Perspective
The difference between zinc and chelated zinc lies in delivery, stability, and efficiency rather than the mineral itself. Standard zinc provides the same essential nutrient, but its absorption is more dependent on digestive conditions and dietary factors.
Chelated zinc addresses these limitations by using a protective structure that improves transport and reduces irritation. This makes it particularly useful for consistent, long-term supplementation.
Understanding these differences allows you to choose based on how your body processes nutrients, not just what appears on the label.
Read More: Stress Management Solutions